Teacher
Professional
- Messages
- 2,669
- Reaction score
- 818
- Points
- 113
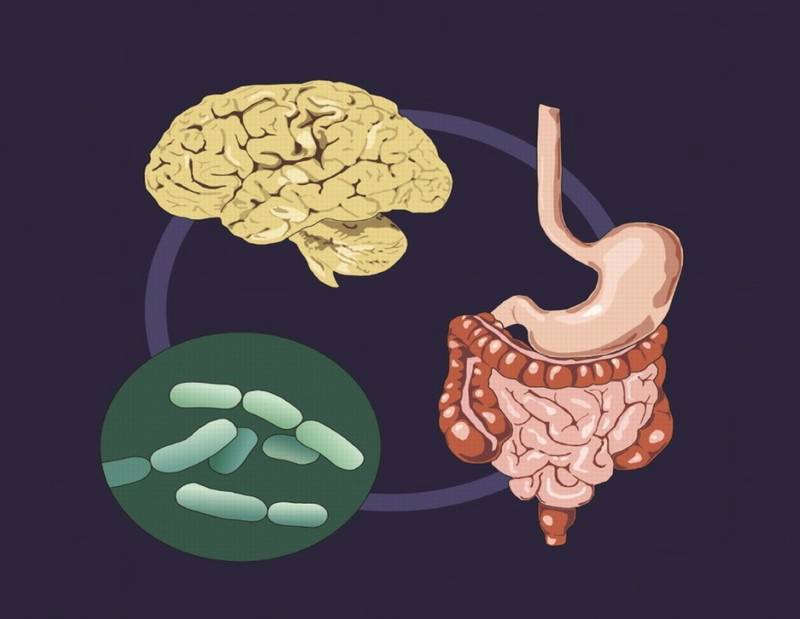
The link between the gut microbiota and the central nervous system is based on a system known as the gut-brain axis. This system is bi-directional. On the one hand, the brain sends signals to the gut that regulate the composition and function of the microbiota. On the other hand, the microbiota interacts with the peripheral nervous system, sending signals through the vagus nerve to the brain, as well as through afferent fibers passing in the sympathetic nerve bundles and innervated by the spinal cord. Afferent fibers of the vagus, distributed throughout the entire thickness of the intestinal wall, are absent in the epithelium and are not in direct contact with the intestinal microbiota. The interaction between microorganisms and the vagus occurs through diffusion of bacterial metabolites and indirectly through the enterochromaffin cells (EECs). EECs interact with the vagus nerve directly through the release of serotonin (5-hydroxytryptamine, 5-HT), which activates 5-HT3 receptors located on vagal afferent fibers and via gut hormones such as cholecystokinin (CCK), glucagon-like peptide-1 ... peptide YY. In addition, EECs perceive signals from the microbiota through toll-like receptors (TLRs), which recognize bacterial lipopolysaccharides, a waste product of microorganisms. However, these are not the only ways in which the gut microbiota and the central nervous system interact. There are also other ways of interaction, EECs perceive signals from the microbiota through toll-like receptors (TLRs), which recognize bacterial lipopolysaccharides, a waste product of microorganisms. However, these are not the only ways in which the gut microbiota and the central nervous system interact. There are also other ways of interaction, EECs perceive signals from the microbiota through toll-like receptors (TLRs), which recognize bacterial lipopolysaccharides, a waste product of microorganisms. However, these are not the only ways in which the gut microbiota and the central nervous system interact. There are also other ways of interaction,
Through the endocrine system, the connection between the microbiota and the central nervous system is realized in two directions, through the hypothalamic-pituitary-adrenal axis, sex hormones and other hormonal systems.
The effect on the hypothalamic-pituitary system was evaluated in a study comparing groups of F344 (stress-sensitive) gnotobionts and pathogen-free rats after stress exposure. The results showed that in gnotobiont rats, serum corticosterone levels were 2.8 times higher than in pathogen-free rats. Also, in gnotobionts, an increase in the expression of corticosterone-releasing hormone receptor mRNA and a decrease in the expression of corticosterone receptor mRNA were noted. These studies have shown that the presence of normal gut microbiota can be considered a protective factor in stress resistance.
In other studies, the use of probiotics Lactobacillus helveticus, Bifidobacterium longum, and prebiotics (fructose and galactose) in depressive-like mice resulted in a decrease in circulating corticosterone levels.
A study conducted on gnotobiont mice with depressive-like behavior showed an increase in urinary cortisol and adrenocorticotropic hormone levels in the blood, while daily administration of probiotics Lactobacillus helveticus, Bifidobacterium longum for 30 days contributed to a decrease in anxiety behavior compared to the control group ... In another part of the study, conducted on human volunteers who took oral probiotics for 30 days, it was shown that the level of anxiety on the HADS scale was reduced.
The results of the study of hypotomical changes showed an increase in the level of corticoliberin and adrenocorticotropic hormone in gnotobionts and pathogenic mice. At the same time, pathogenic mice showed more anxious behavior than gnotobionts, which contradicts previous studies.
A study conducted by Australian scientists on humans found a positive correlation between fecal levels of isovalent acid, a metabolite of the gut microbiota, depression, and the mean morning and midday salivary cortisol. It has been shown that isovalent acid can penetrate the blood-brain barrier and interfere with the release of neurotransmitters. These correlation patterns, in addition to a potential mechanistic model, indicate a potential causal relationship between depression and isovaleric acid.
The connection between the central nervous system and the intestinal microbiota is also realized through the immune system and is bi-directional. The gut microbiota is closely related to the lymphatic system. These interactions involve complex mechanisms and are poorly understood at the moment. However, there is little literature data investigating these mechanisms. The influence of the immune system on the microbiota is reflected in studies showing the role of the pro-inflammatory cytokine IL-6 in the development of depression. The latest research to support this finding has been done in mice with depressive symptoms. The development of symptoms was accompanied by changes in the intestinal microbiota in the form of a predominance of bacteria of the genus Oscillospira in its composition and a decrease in the bacterial ratio of Firmicutes / Bacteroidetes... The administration of antibodies to IL-6 to mice led to normalization of behavior, a decrease in the number of bacteria of the genus Oscillospira, and restoration of the ratio of bacteria of the type Firmicutes / Bacteroidetes.
In another study, the formation of depressive-like behavior was accompanied by the formation of "stress dysbiosis" in the form of a decrease in the number of bacteria of the Porphyromonadaceae family and an increase in the colonization of the cecum with Citrobacter rodentiumcausing colitis in mice. Additionally, the experimental group showed an increase in the expression of TNF mRNA in the large intestine, an increase in the permeability of the intestinal wall and, as a result of translocation of bacteria from the intestine into the blood, markers of neuroinflammation were also found in the prefrontal cortex (an increase in p38MAPK expression, a decrease in PI3K expression, prototein kinase B (Akt) and a nuclear factor activator). Parts of the mice from the experimental group were injected with antibiotics for 21 days, which led to a reversal of depression-like behavior, a decrease in bacterial translocation, and a decrease in p38MAPK expression.
There is a microbiota-inflammasome hypothesis that shows the link between the microbiota and the central nervous system via NLRP3. NLRP3 are innate immune receptors that are part of the inflammasome, capable of detecting danger signals such as uric acid and extracellular ATP of damaged cells. NLRP3 is produced by many immune cells (including microglial cells, monocytes, granulocytes, epithelial cells, and T and B cells) and is involved in the activation of caspase-1. At the moment, studies are accumulating confirming the role of NLRP3 in stress reactions and the development of depressive symptoms and systemic diseases. The microbiota-inflammasome hypothesis of depression formation suggests that stress is able to activate NLRP3, causing anxiety and depressive behavior,
INFLUENCE OF MICROBIOTA ON THE FORMATION OF THE NERVOUS SYSTEM
In healthy individuals, the normal microbiota is relatively stable and forms a synergistic relationship with the host. Violation of this relationship can have serious consequences and lead to the development of not only gastrointestinal and metabolic, but also mental disorders. For example, bi-directional communication between the microbiota and the central nervous system affects stress reactivity, pain perception.
The microbiota goes through an intensive process of maturation and development throughout life. Microbiota formation occurs in parallel with the development of the nervous system, and they have close critical development windows that are sensitive to damage. Early developmental periods, including childhood and adolescence, are the most dynamic periods of change in microbiota and brain development. Disruptions in the dynamic host-microbiota interaction during critical periods can significantly alter signaling between the brain and the gut, affect health, and increase the risk of developing mental disorders throughout life.
Recently, the gut-brain axis has been viewed as one of the key players in the developmental stages of the nervous system, which is also indicated by the fact that events in the perinatal and postnatal periods during the initial colonization and development of microbiota can affect the formation of functional nerve circuits and synaptic plasticity, and also determine not only general, but also mental health in later life. The dogma that the intrauterine environment and the fetus are sterile before delivery is questioned. Currently, there is evidence that suggests that the formation of the primary microbiota coincides with the stages of the formation of the nervous system and critical periods of the formation of the brain. The presence in the fetus of bacteria such as Escherichia coli, Enterococcus faecium and Staphylococcus epidermidismay result from the translocation of maternal intestinal bacteria through the bloodstream and placenta. In addition, a similarity was found between the microbial profile of the placenta and the oral cavity of the mother.
It is important to note that the developing brain in the prenatal period is subject to both exogenous and endogenous influences. A mother's diet, stress, infections can lead to neurodevelopmental disorders, and increase the risk of developing diseases such as autism, attention deficit hyperactivity disorder (ADHD), and schizophrenia. Experimental studies in rodents further support this hypothesis by demonstrating that exposure to microbial pathogens during specific developmental periods leads to behavioral disturbances, including anxiety-like behavior and impaired cognitive function.
During and shortly after birth, the microbiota of the newborn is formed, the source of which is the mother's microbiota. The initial colonization of the intestine depends on the mode of delivery. While babies born in physiological births are colonized by fecal and vaginal bacteria from the mother, babies born by caesarean section are exposed to microorganisms from other biotopes, mainly the mother's skin and the hospital environment. The formation of microbiota in the early stages of development depends on a variety of factors, such as gestational age, type of feeding. The microbiota in formula-fed infants is more diverse than in breastfed infants, in whom the microbiota has a more stable pattern of colonization. However, breastfed babies
INFLUENCE OF MICROBIOME ON NEUROMEDIATORS
The scientific literature discusses the role of the microbiota in the regulation of the serotonergic system. And a study in mice has shown that male gnotobiont pups have increased plasma tryptophan levels, compared with the control group with normal intestinal microbiota. Tryptophan levels returned to normal upon colonization of the intestine by representatives of the normal microbiota, after weaning. Changes in the central nervous system were expressed in male gnotobionts in the form of an increase in the concentration of serotonin in the hippocampus, which did not decrease with normalization of the tryptophan level. An increase in the turnover (circulation) of serotonin in the striatum in gnotobionts was confirmed by determining the ratio of 5-hydroxyindoleacetic acid (a metabolite of serotonin) / serotonin. It is believed that the increase in serotonin levels is associated with enterochromatophin cells.
Other studies also show increased plasma tryptophan and serotonin levels in gnotobiont mice. However, the increase in serotonin and tryptophan levels may be temporary, as shown in one study, where the decrease in serotonin levels in gnotobiont mice occurred on the fourth day, after colonization of the intestines. It should also be noted a study in which a decrease in the concentration of serotonin in the hippocampus was revealed in gnotobiont rats, but an increase in the concentration of serotonin and 5-hydroxyindoleacetic acid, caused by stress, compared with rats with normal intestinal microbiota ... It is not possible to substantiate the differences between mice and rats with gnotobionts, since studies on rats have begun recently.
The direct effect of the intestinal microbiota on the serotonergic system is carried out by limiting the availability of tryptophan for the host organism due to its need for the growth of certain strains and, subsequently, indole synthesis. The presence of the enzyme tryptophan kinase, in some microorganisms, also allows you to synthesize tryptophan on their own.
A decrease in dopamine metabolites, vanillic acid and dihydroxyphenylacetic acid, in patients with depression, indicated a decrease in the intensity of dopaminergic processes. This pattern has been identified in previous studies.
The ability of microorganisms to synthesize GABA has been studied for a long time, but there are few studies showing the ability of representatives of normal intestinal microbiota to synthesize this neurotransmitter. Available data indicate the presence of this ability in some species of bifido and lactobacilli. In the work of Japanese authors, the differences in the level of GABA in the blood, intestinal lumen and in the brain were studied between gnotobiont mice and mice, which, being first gnotobionts, were colonized by the microbiota of ordinary mice. The level of GABA in blood and intestines was significantly higher in the second group. However, no differences were observed in the amount of GABA in the brain in the two groups of mice. This confirms the fact that GABA is synthesized by the intestinal microbiota and enters the bloodstream; and penetration into the brain is strictly controlled by the blood-brain barrier. Most likely, the effect of GABA on the host organism is carried out differently, indirectly, for example, through the vagus nerve or the enteric nervous system. The role of the vagus nerve in the influence of the GABA-synthesizing microbiota was shown in a study where first the introduction of the strain L. rhamnosus JB-1 led to an increase in the number of GABAergic receptors in the brain and to a decrease in anxiety and depression-like behavior compared with the control group. Then, the experiment was repeated with vagotomized mice, ie with mice with a dissected vagus nerve. As a result, administration of L. rhamnosus JB-1 strain to vagotomized mice had no effect on either the expression of GABA receptors in the brain or behavior.
Source: Neznanov NG, Leonova LV, Rukavishnikov GV, Kas'yanov ED, Mazo GE Intestinal microbiota as an object of study in mental disorders. Advances in physiological sciences. 2021. T. 52. No. 1. S. 64-76.